
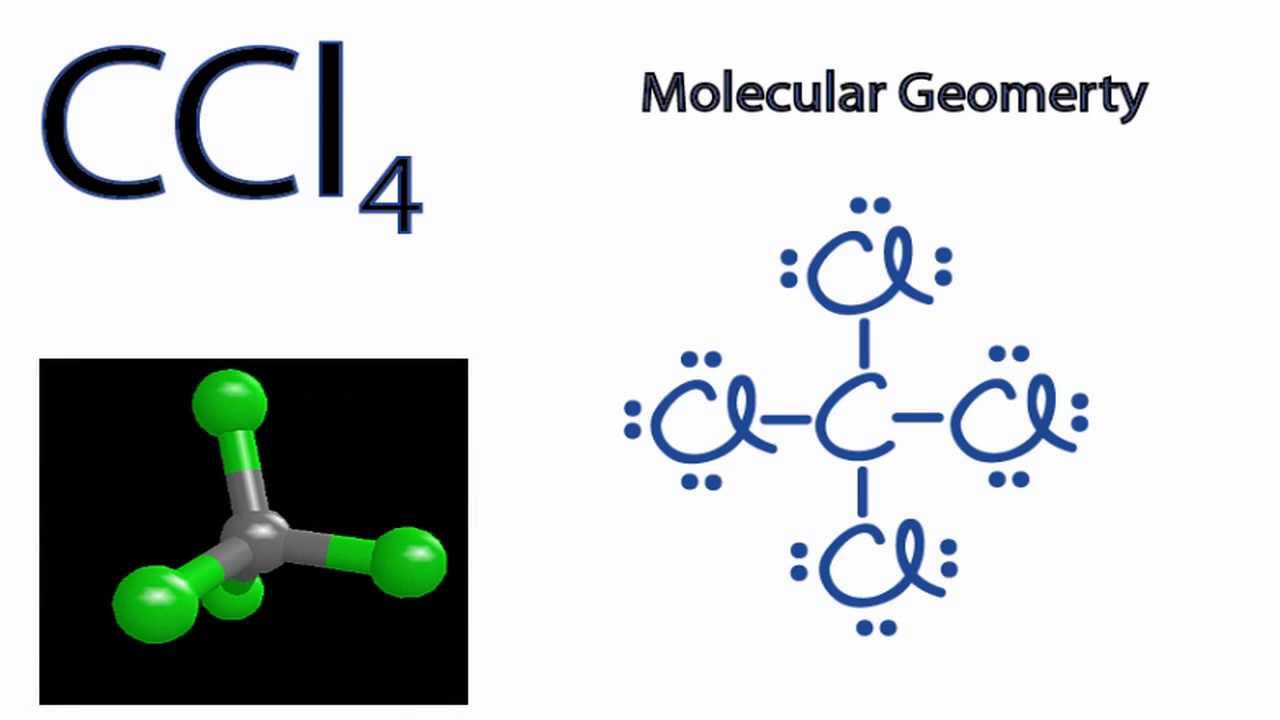
Use The Vsepr Theory To Predict The Shape Of Carbon Tetrachloride Ccl4 A Tetrahedral B Bent C Trigonal Pyramidal D Trigonal Planar Study Com

How Many Pairs Of Electrons Are Shared By The Carbon Atom With Each Chlorine Atom In The Formation Of Carbon Tetrachloride Enotes Com

Carbon Has Four Electrons In The Valence Shell How Does It Attain Stable Electronic Configuration Draw Science Carbon And Its Compounds 11149731 Meritnation Com

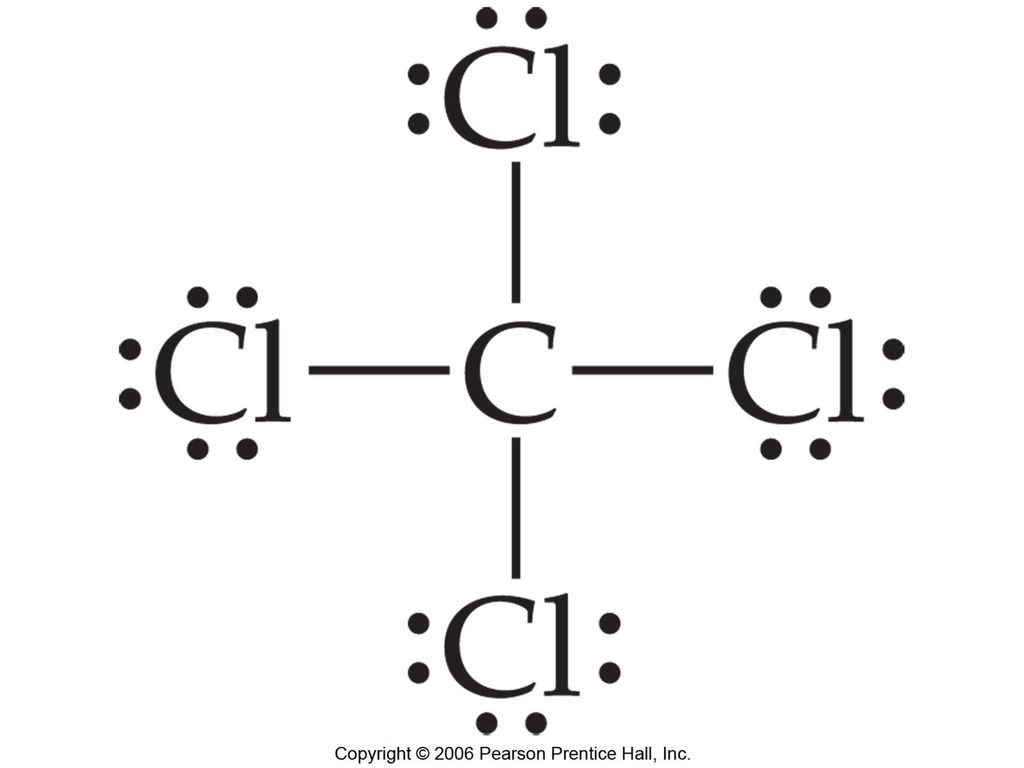
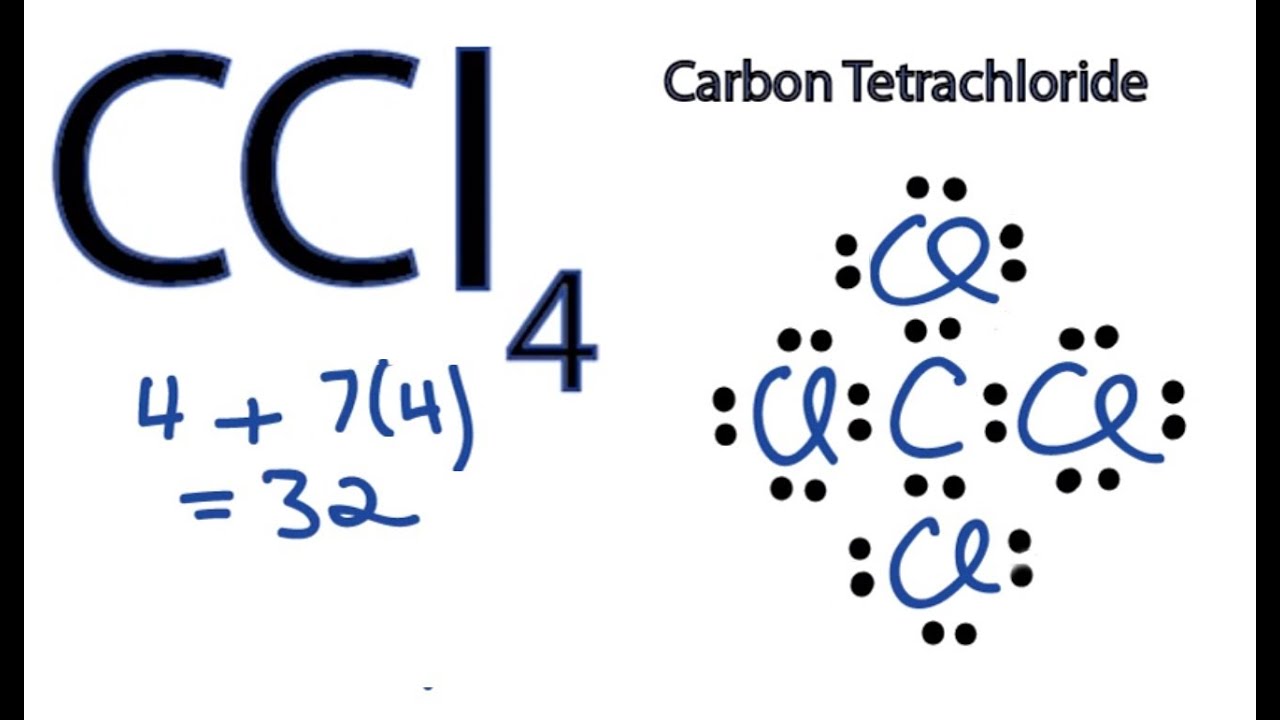
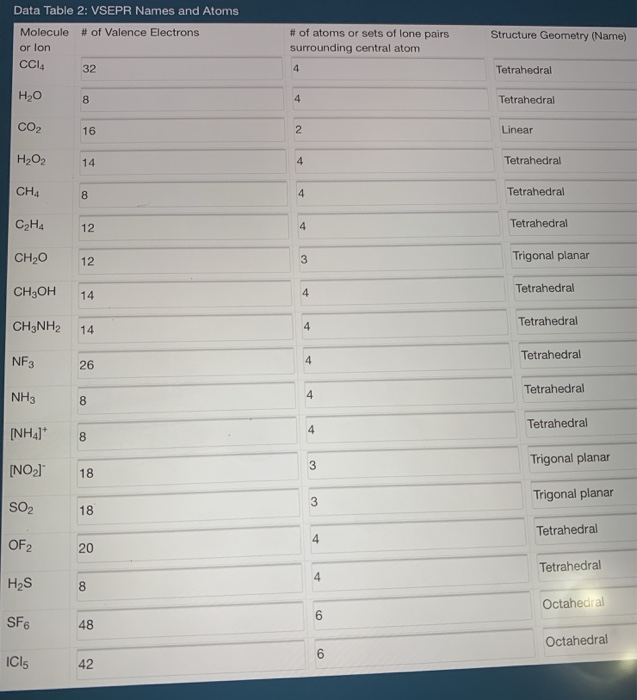
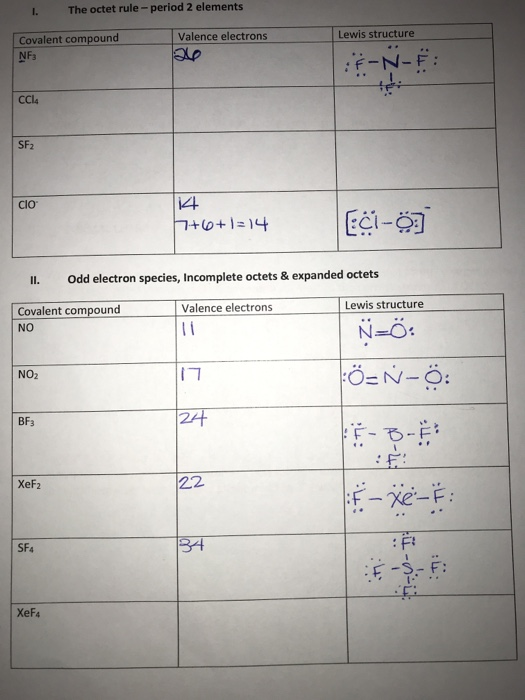
Construct The Lewis Structure Model For The Covalent Compound Carbon Tetrachloride Ccl4 Using The Following Steps 1 The Total Number Of Valence Electrons In Ccl4 Is 2 Write The Atomic Core

The Necessary Steps Required To Show The Formation Of Ccl4 By Lewis Electron Dot Diagram Has Been Jumbled Arrange Them In A Sequence A Thus An Electron Pair Is Shared Between C And










No comments:
Post a Comment